Remote Clinical Trial Monitoring Tools Market Size 2026-2030
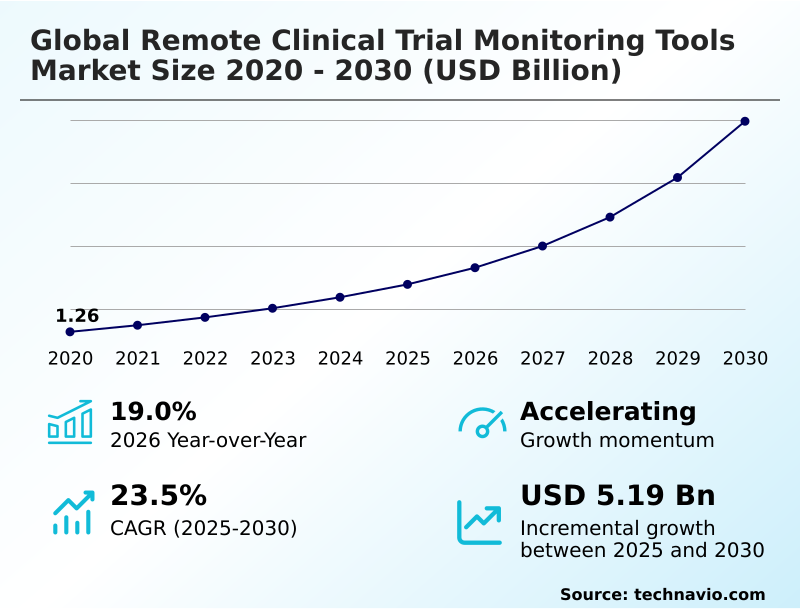
The remote clinical trial monitoring tools market size is valued to increase by USD 5.19 billion, at a CAGR of 23.5% from 2025 to 2030. Rise of decentralized and hybrid clinical trial models will drive the remote clinical trial monitoring tools market.
Major Market Trends & Insights
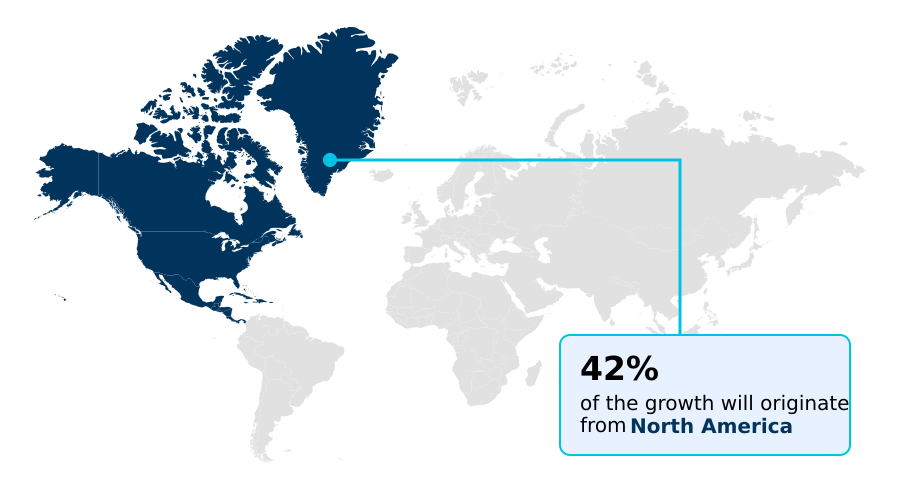
- North America dominated the market and accounted for a 41.7% growth during the forecast period.
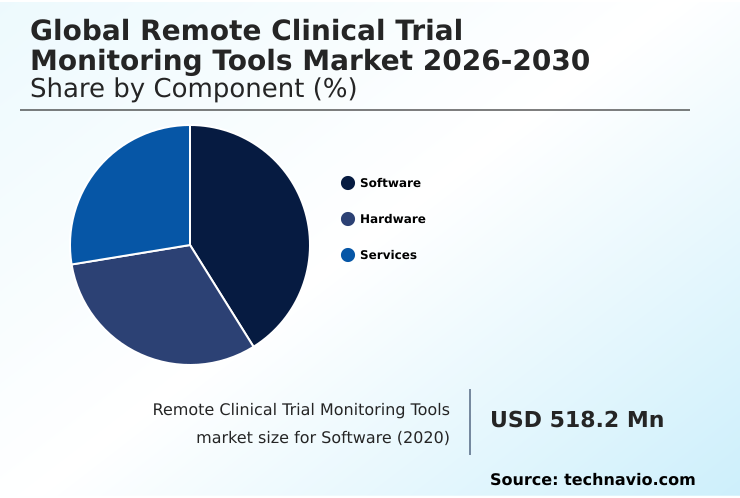
- By Component - Software segment was valued at USD 1.00 billion in 2024
- By End-user - Pharmaceutical and biotech companies segment accounted for the largest market revenue share in 2024
Market Size & Forecast
- Market Opportunities: USD 6.70 billion
- Market Future Opportunities: USD 5.19 billion
- CAGR from 2025 to 2030 : 23.5%
Market Summary
- The remote clinical trial monitoring tools market is undergoing a significant transformation, moving away from traditional, site-intensive oversight toward more agile, technology-driven methodologies. This evolution is propelled by the need for greater efficiency in drug development and the increasing complexity of global studies.
- Core to this shift is the adoption of integrated platforms that combine electronic data capture, real-time analytics, and automated reporting. A key trend is the use of wearable devices and mobile health applications, which provide a continuous stream of patient data, enabling proactive safety monitoring and a deeper understanding of treatment effects.
- For instance, a contract research organization managing a multi-regional oncology trial can leverage a centralized monitoring dashboard to track adverse events across continents in real time, allowing for immediate intervention and ensuring patient safety without the delays of physical travel. However, the industry grapples with challenges related to data interoperability between disparate systems and ensuring robust cybersecurity across decentralized networks.
- Regulatory bodies are increasingly supportive, providing guidance that encourages risk-based approaches, which these tools are uniquely positioned to facilitate, ultimately accelerating the delivery of new therapies.
What will be the Size of the Remote Clinical Trial Monitoring Tools Market during the forecast period?
Get Key Insights on Market Forecast (PDF) Request Free Sample
How is the Remote Clinical Trial Monitoring Tools Market Segmented?
The remote clinical trial monitoring tools industry research report provides comprehensive data (region-wise segment analysis), with forecasts and estimates in "USD million" for the period 2026-2030, as well as historical data from 2020-2024 for the following segments.
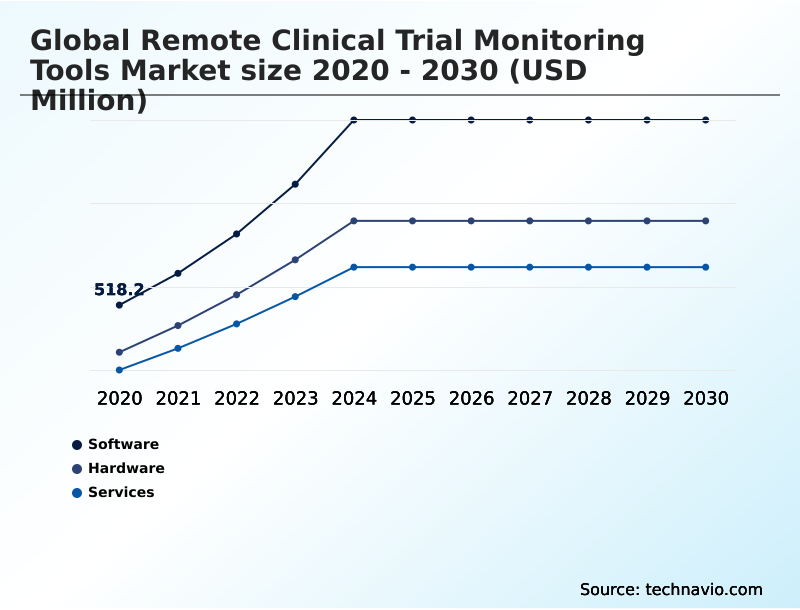
- Component
- Software
- Hardware
- Services
- End-user
- Pharmaceutical and biotech companies
- Contract research organizations
- Healthcare IT and service providers
- Academic and research institutes
- Deployment
- Cloud based
- On premises
- Geography
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Asia
- Rest of World (ROW)
- North America
By Component Insights
The software segment is estimated to witness significant growth during the forecast period.
The software segment is foundational, providing the architectural backbone for modern clinical research. These platforms are shifting from simple electronic data capture systems to integrated suites that manage the entire trial lifecycle.
Key functionalities include electronic clinical outcome assessments and the collection of patient reported outcome measures, which are essential for patient-centric trial designs.
Leading clinical trial management system offerings now embed real-time data analytics and automated data reconciliation, enabling a 40% reduction in data cleaning timelines.
The evolution of clinical data management software focuses on digital health technology integration and ensuring the interoperability of eclinical systems.
This transition to sophisticated remote patient monitoring services and pharmacovigilance reporting tools supports a more agile and data-driven approach to clinical trial workflow automation and eRegulatory document management.
The Software segment was valued at USD 1.00 billion in 2024 and showed a gradual increase during the forecast period.
Regional Analysis
North America is estimated to contribute 41.7% to the growth of the global market during the forecast period.Technavio’s analysts have elaborately explained the regional trends and drivers that shape the market during the forecast period.
See How Remote Clinical Trial Monitoring Tools Market Demand is Rising in North America Request Free Sample
North America leads market adoption, driven by a mature digital health infrastructure and supportive regulatory frameworks that encourage the use of cloud-based clinical platforms.
This region is a hub for innovation in real-time site performance tracking and digital health technology validation, with many organizations implementing unified clinical trial platforms for multi-regional trial management.
The emphasis on trial data integrity has spurred the development of advanced centralized monitoring dashboards and robust clinical data repository solutions.
These systems, supported by sophisticated biostatistical analysis software, allow sponsors to analyze performance metrics with high precision, improving site performance management tools and clinical study protocol design.
European markets are also advancing, focusing on pharmacovigilance process management and deploying eSource data capture to meet stringent data privacy standards.
Market Dynamics
Our researchers analyzed the data with 2025 as the base year, along with the key drivers, trends, and challenges. A holistic analysis of drivers will help companies refine their marketing strategies to gain a competitive advantage.
- The strategic implementation of remote clinical trial monitoring tools is reshaping research paradigms, with a strong focus on enhancing both efficiency and data quality. The use of real time patient safety monitoring tools has become a cornerstone of modern trial design, particularly in high-risk therapeutic areas.
- A pivotal development is the application of AI in clinical trial data verification, where algorithms can identify discrepancies with twice the speed of manual review, significantly improving clinical trial data integrity. Decentralized trial data collection methods, which heavily rely on patient-reported outcomes via mobile apps, are now standard, enabling broader and more diverse recruitment.
- This is especially impactful in remote monitoring for oncology trials, where patient-centricity is paramount. Key technical challenges, such as ensuring secure data transmission in clinical research and the interoperability of eclinical systems, are being addressed through advanced cloud based electronic data capture and standardized protocols. Implementing a risk based monitoring strategy implementation is critical for reducing clinical monitoring travel costs.
- Firms are now automating source data verification process and integrating EHR data into EDC systems to streamline the clinical research associate workflow. This digital shift helps in managing multi-regional clinical trials effectively while ensuring GDPR compliance in trials.
- Predictive analytics for trial risks and tools for improving patient retention with remote tools further optimize trial execution, leading to faster regulatory submissions.
What are the key market drivers leading to the rise in the adoption of Remote Clinical Trial Monitoring Tools Industry?
- The primary market driver is the rising adoption of decentralized and hybrid clinical trial models, which necessitate advanced remote oversight.
- The primary driver is the industry-wide adoption of decentralized clinical trial models and virtual clinical trial solutions, which enhance patient recruitment and retention.
- This shift is enabled by regulatory compliance automation and a strong emphasis on a risk-based monitoring approach, which focuses resources on high-risk areas, improving efficiency by up to 40%.
- The use of electronic informed consent and remote imaging review streamlines investigative site workflows and ensures good clinical practice compliance. Furthermore, the implementation of risk-based quality management and automated query management reduces manual oversight.
- This digital transformation is critical for clinical operations optimization and supports timely regulatory submission support by ensuring patient safety indicators are monitored continuously.
What are the market trends shaping the Remote Clinical Trial Monitoring Tools Industry?
- A key market trend is the integration of predictive analytics and machine learning, which shifts monitoring activities from a reactive to a proactive model.
- A significant trend is the shift toward proactive oversight through the integration of a predictive analytics module and AI-driven data cleaning. This enables the analysis of high-frequency data streams from wearable biosensor integration, providing deeper insights for real-world evidence generation.
- These systems utilize natural language processing verification and advanced clinical data visualization to identify anomalies, improving data accuracy by over 25%. The adoption of a digital biomarker platform and continuous patient monitoring through patient engagement platforms supports this trend.
- As trial data interoperability improves, ePRO data collection via telehealth integration for trials is becoming seamless, allowing for more comprehensive and timely participant feedback.
What challenges does the Remote Clinical Trial Monitoring Tools Industry face during its growth?
- A key challenge affecting industry growth stems from significant data security and cybersecurity vulnerabilities inherent in remote data transmission.
- A critical challenge is ensuring clinical trial data security across fragmented digital ecosystems. The reliance on cloud data storage for trials and remote access necessitates robust secure data transmission protocols, multi-factor authentication protocols, and comprehensive trial data access control.
- The lack of standardized clinical data interchange standards complicates the integration of disparate eClinical solution suite platforms, leading to manual source data verification automation and data reconciliation efforts. Organizations must manage complex investigative site workflows and ensure electronic source documentation is protected with features like data anonymization features.
- This requires significant investment in security infrastructure to manage eCOA data management and protect against breaches, which can increase operational costs by 15-20%.
Exclusive Technavio Analysis on Customer Landscape
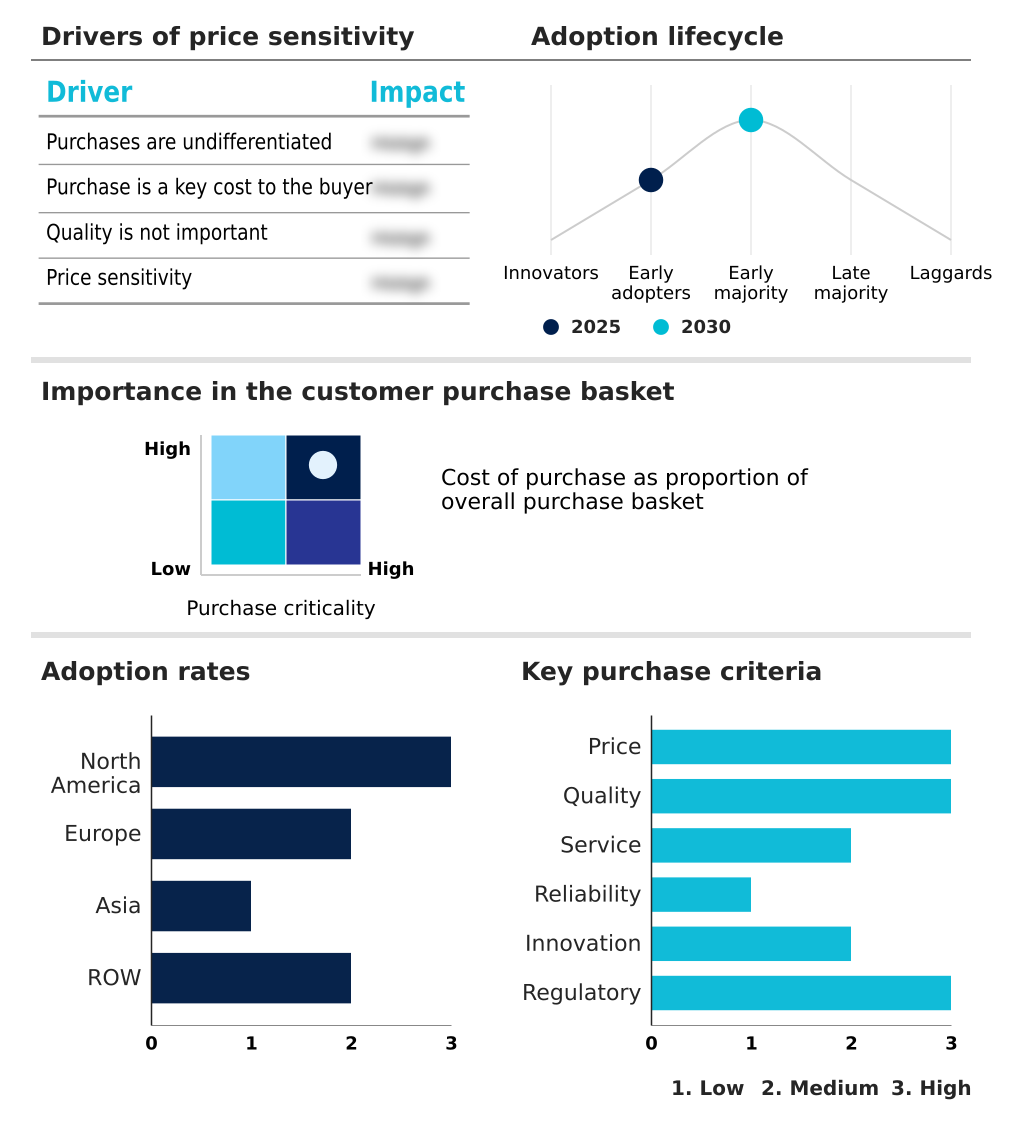
The remote clinical trial monitoring tools market forecasting report includes the adoption lifecycle of the market, covering from the innovator’s stage to the laggard’s stage. It focuses on adoption rates in different regions based on penetration. Furthermore, the remote clinical trial monitoring tools market report also includes key purchase criteria and drivers of price sensitivity to help companies evaluate and develop their market growth analysis strategies.
Customer Landscape of Remote Clinical Trial Monitoring Tools Industry
Competitive Landscape
Companies are implementing various strategies, such as strategic alliances, remote clinical trial monitoring tools market forecast, partnerships, mergers and acquisitions, geographical expansion, and product/service launches, to enhance their presence in the industry.
Advarra Inc. - Offerings center on integrated eRegulatory and electronic data capture platforms, enabling real-time site performance tracking and document management for streamlined remote trial oversight.
The industry research and growth report includes detailed analyses of the competitive landscape of the market and information about key companies, including:
- Advarra Inc.
- Anju Software Inc.
- Castor Research Inc.
- Cloudbyz Inc.
- Ennov SAS
- Florence Healthcare.
- IQVIA Holdings Inc.
- Medable Inc.
- Medidata
- Medrio Inc.
- Merative L.P.
- Microsoft Corp.
- Obvio Health USA Inc.
- OpenClinica LLC
- Oracle Corp.
- Salesforce Inc.
- SAP SE
- Signant Health
- Veeva Systems Inc.
- Zoom Video Communications Inc.
Qualitative and quantitative analysis of companies has been conducted to help clients understand the wider business environment as well as the strengths and weaknesses of key industry players. Data is qualitatively analyzed to categorize companies as pure play, category-focused, industry-focused, and diversified; it is quantitatively analyzed to categorize companies as dominant, leading, strong, tentative, and weak.
Recent Development and News in Remote clinical trial monitoring tools market
- In August 2024, the European Medicines Agency published a comprehensive reflection paper detailing technical standards for validating remote monitoring software using artificial intelligence, reducing implementation uncertainty for sponsors.
- In October 2024, a leading clinical software technology provider introduced a machine learning module using natural language processing to automate the reconciliation of remote source data, eliminating manual CRA intervention.
- In March 2025, a global provider of clinical trial software launched a specialized analytics module that employs neural networks to predict site-level enrollment delays and potential protocol deviations.
- In May 2025, Oracle Life Sciences announced a major update to its clinical software platform, integrating generative AI to automate the initial validation of patient data, processing information 50% faster.
Dive into Technavio’s robust research methodology, blending expert interviews, extensive data synthesis, and validated models for unparalleled Remote Clinical Trial Monitoring Tools Market insights. See full methodology.
| Market Scope | |
|---|---|
| Page number | 294 |
| Base year | 2025 |
| Historic period | 2020-2024 |
| Forecast period | 2026-2030 |
| Growth momentum & CAGR | Accelerate at a CAGR of 23.5% |
| Market growth 2026-2030 | USD 5185.6 million |
| Market structure | Fragmented |
| YoY growth 2025-2026(%) | 19.0% |
| Key countries | US, Canada, Mexico, Germany, UK, France, Italy, The Netherlands, Spain, Russia, China, India, Japan, South Korea, Indonesia, Thailand, Singapore, Australia, UAE, Brazil, South Africa, Saudi Arabia and Turkey |
| Competitive landscape | Leading Companies, Market Positioning of Companies, Competitive Strategies, and Industry Risks |
Research Analyst Overview
- The remote clinical trial monitoring tools market is defined by a rapid shift from periodic, manual oversight to continuous, automated surveillance. This evolution is underpinned by the convergence of decentralized clinical trial models and the risk-based monitoring approach, which together optimize resource allocation.
- The integration of a predictive analytics module and real-time data analytics into cloud-based clinical platforms enables the proactive identification of issues related to trial data integrity. For board-level consideration, the adoption of a digital biomarker platform directly influences strategic investment, as it can de-risk clinical programs by providing objective endpoints, reducing trial timelines by up to 30%.
- Key technologies such as wearable biosensor integration and remote source data verification are no longer novelties but essential components for maintaining good clinical practice compliance.
- The market's trajectory is toward comprehensive solutions that manage everything from electronic clinical outcome assessments and patient reported outcome measures to pharmacovigilance process management and automated data reconciliation, all facilitated by robust clinical data interchange standards and electronic data capture systems.
What are the Key Data Covered in this Remote Clinical Trial Monitoring Tools Market Research and Growth Report?
-
What is the expected growth of the Remote Clinical Trial Monitoring Tools Market between 2026 and 2030?
-
USD 5.19 billion, at a CAGR of 23.5%
-
-
What segmentation does the market report cover?
-
The report is segmented by Component (Software, Hardware, and Services), End-user (Pharmaceutical and biotech companies, Contract research organizations, Healthcare IT and service providers, and Academic and research institutes), Deployment (Cloud based, and On premises) and Geography (North America, Europe, Asia, Rest of World (ROW))
-
-
Which regions are analyzed in the report?
-
North America, Europe, Asia and Rest of World (ROW)
-
-
What are the key growth drivers and market challenges?
-
Rise of decentralized and hybrid clinical trial models, Data security and cybersecurity vulnerabilities
-
-
Who are the major players in the Remote Clinical Trial Monitoring Tools Market?
-
Advarra Inc., Anju Software Inc., Castor Research Inc., Cloudbyz Inc., Ennov SAS, Florence Healthcare., IQVIA Holdings Inc., Medable Inc., Medidata, Medrio Inc., Merative L.P., Microsoft Corp., Obvio Health USA Inc., OpenClinica LLC, Oracle Corp., Salesforce Inc., SAP SE, Signant Health, Veeva Systems Inc. and Zoom Video Communications Inc.
-
Market Research Insights
- The market's momentum is driven by the strategic adoption of virtual clinical trial solutions and unified clinical trial platforms that enhance operational efficiency. Organizations are leveraging eClinical solution suites to streamline workflows, achieving up to a 35% reduction in study setup times.
- The focus on real-world evidence generation is intensifying, with patient engagement platforms facilitating better data collection and improving retention rates by over 20% compared to traditional methods. Furthermore, the implementation of robust clinical trial data security measures is paramount.
- Advanced trial data access control and secure data transmission protocols are becoming standard, addressing critical regulatory compliance automation needs while supporting complex clinical operations optimization. This shift toward integrated digital health technology integration ensures a more cohesive and secure research environment.
We can help! Our analysts can customize this remote clinical trial monitoring tools market research report to meet your requirements.


 RIA -
RIA - 
